So. These past few months of grade 9 have been…… busy. Of course we have done tons of humanities and maker classes and lessons which you have seen in my posts but, what you haven’t heard much about yet is… scimatics!!
Scimatics is our science and math class combined into one course that we take everyday. Sometimes we do science and sometimes we do math, it depends on the day or more like month, I guess. Before this mystery unit that I am about to ell you about, we have did electricity and circuits which I talked a bit about in a post called Metaphor Machines. But now, for the big reveal. The latest and greatest unit we have been working on is….. CHEMISTRY!!
Most people who haven’t done chemistry before just assume it what they show int he movies. You pour to colourful liquids into a cup and then bam! You have an explosion! But no. Chemistry is much much more than that. It has to do with atoms, ions, neutrons, protons, electrons, and so much more. Most of that may not make any sense to you but don’t worry because I will explain and it will start to make more sense throughout the post. Here is the dictionary definition of chemistry.
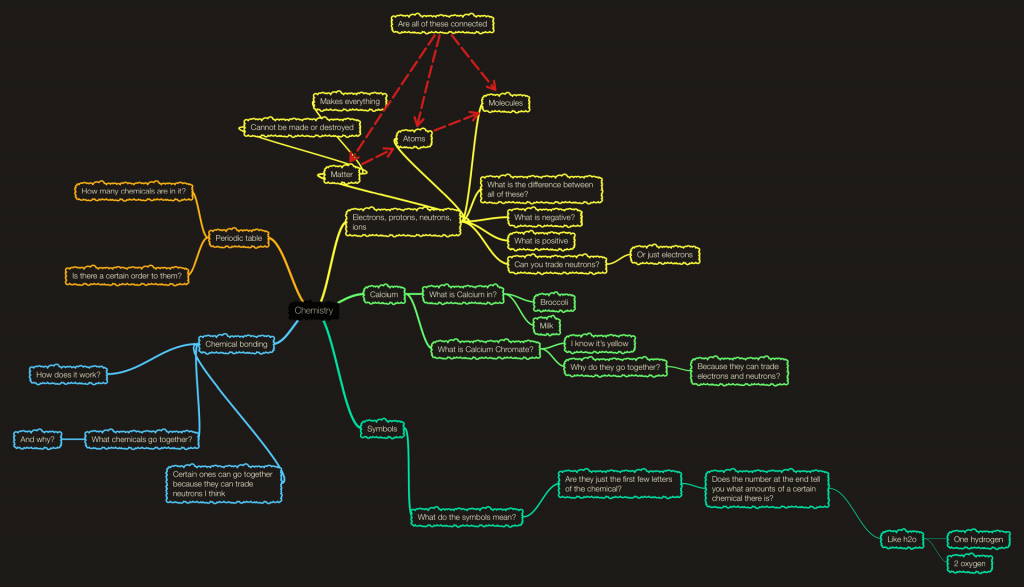
That definition took a lot of classes of learning and thinking before I could get to what I know today, which still is only a small part of chemistry. But everyone has to start somewhere, and I started with a mind map. If you don’t know what a mins map is, check out my post called what-ology and there is a good section int here explaining it. Anyway, at the beginning of each science unit we do a mind map showing what we know which usually isn’t much. After the unit is over, we add to the original one but with all of the information we know now! It’s pretty cool. Here is my unit start mind map:
With that information, I set off on working on my first project, a project our class likes to call, Chem Stories. Without knowing it, this project was for something much bigger than we thought which you will here about in a future post, it is TBA. My partner was Gabby, we have the same last name, it’s pretty cool, and our task was to create an animated video using an app called Keynote to show ionic and covalent bonding.
Now after watching that video, you are still probably very confused about the whole bonding deal so here I am to explain it to you. That video followed the path of Spike Strontium and Olive Oxygen which in real life, are actually elements on the periodic table!
And getting even more specific, they even have special little diagram that show what the atom looks like called Bohr diagrams.
IONIC BONDING
Now, if you look at those two Bohr diagrams, do you notice anything? Maybe not, but I definitely do. I notice that strontium has one ring with only two electrons on it. That outside ring is called the valence ring or shell and the little dots on it are called valence electrons, every atom has a valence ring with a certain amount of electrons on it. All of the other dots on the outside rings are also electrons. The red and blue dots in the centre is what we call the nucleus, like the brain of the atom and it is made up of protons and neutrons. That’s a little bit confusing so here is an annotated atom so y’all can understand a bit better.
Now, since Strontium only has two electrons on his outside ring, that is kind of useless, he really wants to get rid of those, leave them in dust. But he can’t just throw them in the trash can, he as to find someone to bond with. Someone that can take his extra electron and you know who is perfect for that? OXYGEN! Oxygen has six valence electrons, and I may not have mentioned this before, but the ideal number for valence electrons is 8. So if strontium wants to get rid of those two and oxygen has room for two, Strontium can just transfer them over to oxygen. That is called an ionic bond.
One more important piece of information is that since strontium had two extra electrons, he has an over all atomic charge of 2 positive and oxygen, since she is missing two, is two negative. So when they do the transfer they become a neutral ionic compound, they have a charge of, well, nothing! They are right in the middle. A condition of transferring electrons, as you saw in our video, is that one that happens, the two elements are bonded, stuck together forever! The bond between strontium and oxygen is ionic because strontium is a metal and ionic bonds allow metals and non metals to be involved in the process, creating a chemical we like to call, strontium oxide!
COVALENT BONDING
As well as an ionic bond, there is also something we call a covalent bond which you saw in our video as well. This happened between Harry Hydrogen and Flory Fluorine and it was a little bit different than the way Spike and Olive bonded. We know that they literally transferred electrons to become complete but there is actually another way another way. First things first, let’s take a look at the Bohr models for these two elements.
So, Hydrogen only has one valence electron which is nowhere near 8 and fluorine is only one away! So instead of transferring, they can actually just attach and share electrons making sure that they both have 8! That would look a little something like this and be called hydrogen fluoride.
CONCLUSION
After all of that learning plus more terms such as these (I made this poster in Canva):
I made a unit end mind map!
As you can see it is much larger than the original mind map. It’s so cool to look back and see the questions I asked, to see how confusing it was and now I can answer my OWN questions easily. Chemistry is probably one of my favourite topics in science right now, and what mad exit even better was making our learning into a video, it helped me understand a lot better when I actually gave the elements personalities and traits. So, I can’t wait to continue chemistry throughout the years because it is so much fun!
~Your blogging friend, Jordyn















Leave a Reply